Experimental Neuro-Oncology
(Team Leader: Attila Németh)
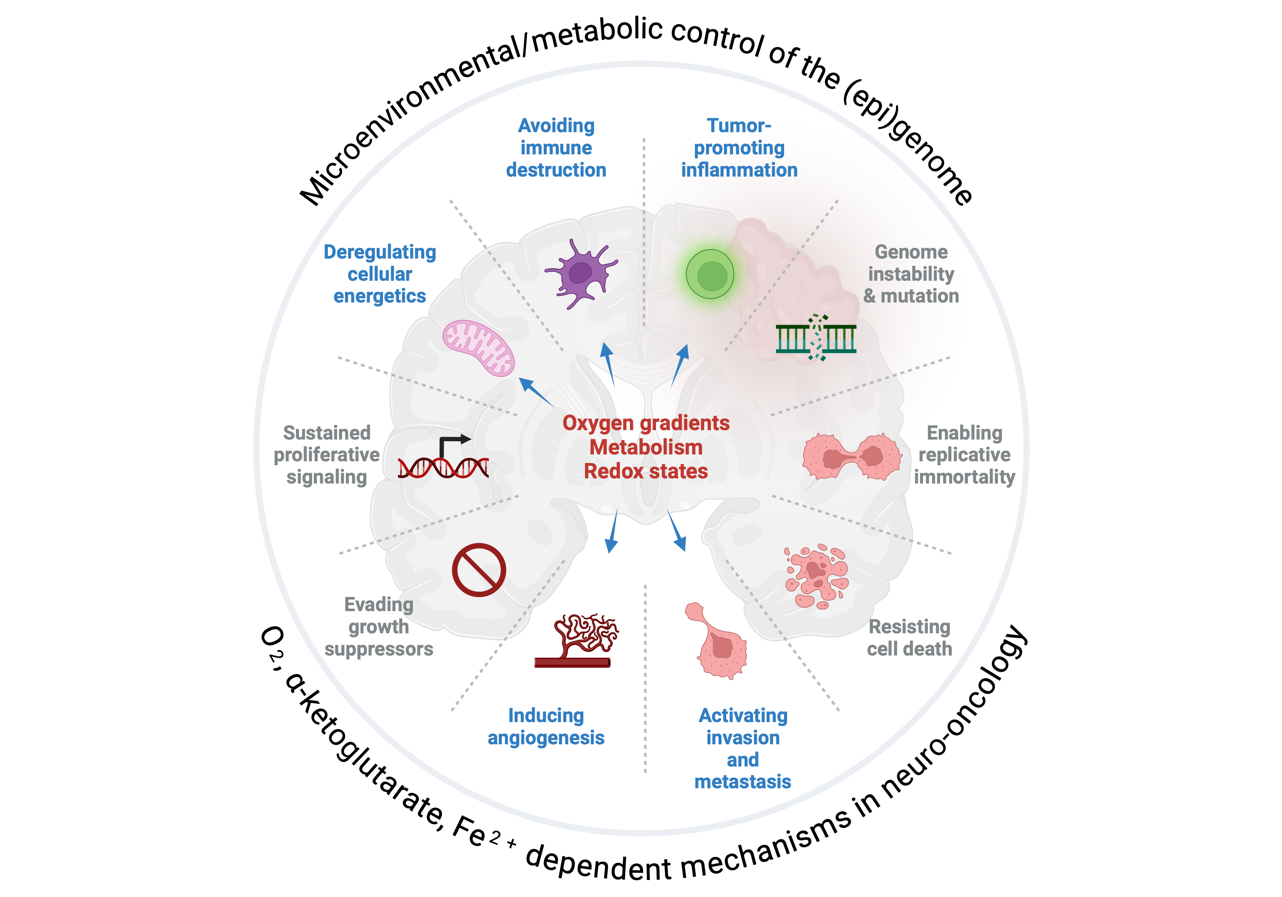
The focus area Experimental Neuro-Oncology seeks to understand how tumor cells perceive and adapt to microenvironmental stressors such as hypoxia and metabolic fluctuations, which are critical for driving key hallmarks of cancer, including angiogenesis, immune evasion, and metastasis. A particular focus is placed on studying microenvironmental interactions during primary tumor growth and tumor colonization/metastasis, both within the brain and in the lung. These interactions shape tumor behavior, influencing progression, metastasis, and therapy resistance.
We aim to uncover how stress signals from the tumor microenvironment dynamically reprogram the metabolic and epigenetic states of cancer cells, enabling their survival and adaptability in diverse and hostile conditions. By linking these fundamental processes to translational efforts, we aim to identify actionable therapeutic targets that address therapy resistance. Our research integrates cutting-edge experimental and computational approaches, leveraging patient-derived data to enhance the clinical relevance of our findings.